- Blog
- Retro video filter
- Download marvel ultimate alliance gold edition for pc
- Lingua latina per se illustrata answer key pdf
- New fairy tail episodes
- Why does toontown rewritten keep crashing
- Ilayaraja telugu songs download
- Meggan grubb girls with muscle
- Rpg maker controls pc
- Call of duty 4 mpdata level 55 download
- Castleminer z codes
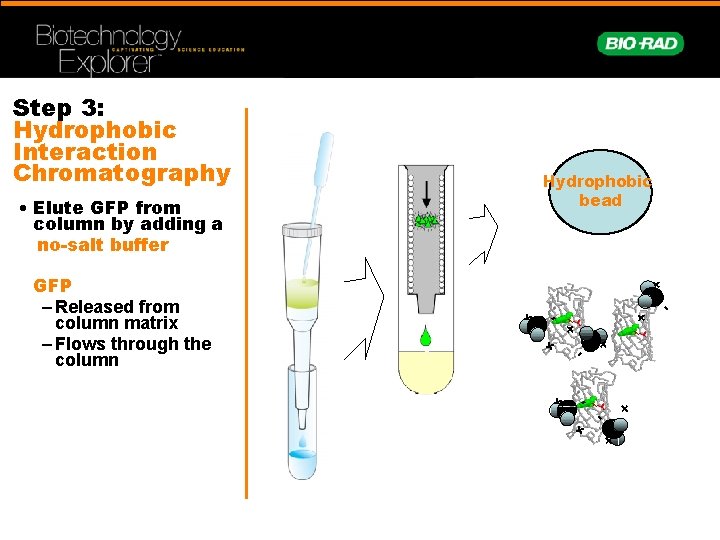
- Gfp hydrophobic amino acids
- Play extensions games city car driving activation key
- Teori produksi ekonomi mikro sadono sukirno
- Gundham x sonia
- Review butch vig vocals
- Nuclear scaffold drawing
- Opengl 4-4 download
- Descargar gta san andreas
- Mii channel theme funny
- What is gracie master cycle
- Viki sky castle
- Bgg games like dark souls
- Gnome stronghold fairy ring
- Digidesign digi 003 factory pro tools le workstation
- Windows 7 internet usage monitor
- Steam iw3mp-exe has stopped working
- How much does ironcad cost
- Nero 2014 platinum updates
- Duniyadari marathi movie free online
Wild type GFP has a number of interesting characteristics that can potentially complicate its applicability to biosensing.One is its tendency to aggregate in the cell, especially when expressed in high concentrations. Figure 2 depicts these stabilizing hydrogen bonding interactions with the chromophore.Additionally, a number of internal residues interact with and stabilize Arg96, a side chain that is known to be required for the maturation of the chromophore.Specifically, Thr62 and Gln183 form hydrogen bonds with the protonated form of Arg96 stabilizing a buried positive charge within the GFP beta barrel, which in turn stabilizes a partial negative charge on the carbonyl oxygen of the imidazolidone ring.ġ.2. The interior of the GFP beta barrel is unusually polar.There is an interior cavity filled with four water molecules on one side of the central helix, while the other side contains a cluster of hydrophobic side chains which is more typical of a protein core.Several polar side chains interact with and stabilize the GFP chromophore.Three of these, His148, Thr203, and Ser205, form hydrogen bonds with the phenolic hydroxyl group of the chromophore.Arg96 and Gln94 interact with the carbonyl group of the imidazolidone ring. Numbers indicate the start and end of each secondary structure element.
#Gfp hydrophobic amino acids code
Tertiary structure of GFP as determined by x-ray crystallography (PDB code 2B3P).Shown on the bottom is a cartoon depicting the secondary structure elements, all anti-parallel beta strand pairings except β1 to β6, which is parallel. Therefore, the closed beta barrel structure is essential for fluorescence by shielding the chromophore from bulk solvent. The molecular structure of GFP is illustrated in Figure 1 along with a cartoon representation showing the organization of the secondary structure elements that compose the beta barrel.Each beta strand is 9 to 13 residues in length and hydrogen bonds with adjacent beta strands to create an enclosed structure.The bottom of the barrel contains both termini and two distorted helical crossover segments, and the top has one short crossover and one distorted helical crossover segment.The beta-barrel (sometimes referred to as a “beta can” because it contains a central alpha-helical segment) consists of three anti-parallel three-stranded beta-meander units and a two-stranded beta-hairpin (shown in blue, green, and yellow, and red in Figure 1 respectively).The very distorted central alpha helix contains three residues which participate in an auto-catalyzed cyclization/oxidation chromophore maturation reaction which generates the p-hydroxybenzylidene-imidazolidone chromophore.In the unfolded state, the chromophore is non-fluorescent, presumably because water molecules and molecular oxygen can interact with and quench the fluorescent signal. One of the most obvious features of its tertiary structure is a beta-barrel composed of 11 mostly-antiparallel beta strands. The molecular structure of GFP was first determined in 1996 using X-ray crystallography.
